A technological revolution is providing insights into how the brain associates memories, a process essential to understanding and organizing the world around us
- The study of memory has undergone a revolution: new imaging methods that follow the electrical activity of individual nerve cells and even activate or deactivate cells with precise timing, allow neuroscientists to perform experiments that were considered science fiction just a few years ago.
- Methods currently available to neurobiologists have shown that memories are not randomly assigned to neurons in brain areas related to information processing and storage, but specific mechanisms determine which cells will store the memory.
- The brain's ability to determine which nerve cells will encode which memories is essential for strengthening memories and linking them, characteristics that are affected in many neuropsychiatric diseases and during cognitive decline with age.
Our memories depend on our ability to retrieve from our minds details about the world - a girl's face, a goose, a lake. But to turn them into actual experiences, the mind has to somehow blend these single elements into one whole - the look on the girl's face when she sees a flock of geese suddenly fly out of the nests on the lake shore.
Consolidated memory also relies on other factors. Our survival through the ages has depended on our ability to remember not only the correct information - for example a lion or a snake - but also the context. Did we encounter the animal during a sudden confrontation in an isolated patch of African savannah or as part of a leisurely visit to the San Diego Zoo?
In order to avoid other types of predators in our daily lives, we also need to link memories over time: to know if it is worth making a seemingly attractive investment, it is necessary to take into account the source of the recommendation, for example the fairness of the recommenders. The inability to connect the two things can lead to disastrous results.
Neuroscience is beginning to deal with how the brain links memories across space and time. Until now, most research has focused on how we acquire, store, and retrieve individual memories. However, most memories do not exist as single entities. Instead, one recollection leads to another recollection, so that a tangled sequence of memories is created that help us envision and gain a better understanding of the world around us.
The basic mechanisms the brain uses to produce these linked memories are beginning to emerge, after 20 years of research in my lab and other labs. Understanding the physiological processes involved in interweaving individual memories will not only provide insights into how the brain works, but will also help us prevent memory problems that impair our ability to create and bind memories together.
A successful accident
When we began our research on linking memories together in the late 90s, we did not have the tools and basic knowledge needed to tackle the subject. An essential first step in understanding the fabric of memories was made when we discovered the concept of memory allocation, that is, the fact that the brain uses specific rules to assign pieces of learned information to given groups of nerve cells (neurons) in brain regions involved in memory formation.
Luck played a key role in discovering the allocation of memories. It all started with a conversation between me and Michael Davis, a friend and colleague who now works at Emory University, during a visit to Yale University in 1998. Davis shared with me the findings of studies in which his laboratory changed the so-called gene CREB To strengthen emotional memories in rats - for example the link between a certain sound and an electric shock. In the past, my lab, now at UCLA, and other labs have shown that the CREB gene is required to form long-term memories. The CREB gene fulfills this role by coding for a protein that controls the expression of other genes needed to form memories. During learning, some of the synapses (the cellular structures that nerve cells use to communicate with each other) are built, or strengthened, so that they can encourage connections between cells. The CREB protein acts as a molecular architect of this process. Without his help, most of our experiences would be forgotten.
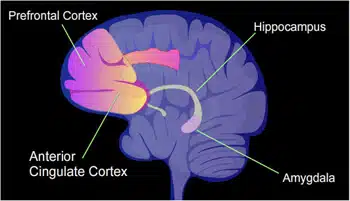
What surprised me was that Davis' group was able to improve memory, even though his lab increased CREB levels in only a small portion of the population of neurons inAmygdala, the same area of the brain essential for emotional memories. The question that accompanied me for months after my visits to Yale was: How did it happen that the memory was only formed in the small number of cells where it could take advantage of the high levels of CREB? Could it be that CREB not only orchestrates memory formation but also helps ensure that cells with CREB are more likely to be involved in memory formation? In our studies of CREB, we focused on its role in certain areas of the brain that we knew were associated with memory: the amygdala andHippocampus, which contains an internal map of our environment.
Science involves finding the right questions just as much as finding the answers. What talking with Davis helped me realize is that neurobiologists knew very little, if any, about the rules by which a given memory is assigned to neurons in each of the brain regions that process and store memories. That's why we decided to check the issue more carefully.
Our first breakthrough came after we recruited neurobiologist Sheena A. Joslin, who was studying CREB in Davis' lab. In a series of animal experiments she conducted in my lab and then with colleagues in her lab at the University of Toronto, Jocelyn used a virus to inject extra copies of CREB into certain neurons in the mouse amygdala. She showed that these neurons were four times more likely to store fearful memories than neighboring neurons.
In 2007, after almost a decade of efforts, my laboratory, in collaboration with Joslin's group, published evidence that emotional memories are not randomly assigned to neurons in the amygdala, but that the cells destined to store these memories have more CREB protein. Further experiments showed that CREB has a similar role in other brain areas, including the hippocampus and the cerebral cortex, the outermost layer of the brain.
Turning memories on and off
To confirm CREB's role in memory allocation, we turned to innovative methods that have changed the face of memory research in recent years. These laboratory methods make it possible to turn nerve cells on or off, and in effect to stimulate or silence a memory.
As one example, Yu Zhu, then in my lab, genetically engineered a small group of mouse amygdala neurons so that they had higher levels of CREB and also expressed another protein that had been genetically engineered in Edward Callaway's lab at the Salk Institute for Biological Studies in La Hoya, California. Callaway's cool protein allowed us to silence CREB neurons at will. When the neurons with the highest levels of CREB were turned off and the neurons with lower levels of the protein remained active, the emotional memories were suppressed, a result that provides evidence that neurons with higher levels of CREB are more likely to be involved in the formation of memories.
We knew that higher levels of CREB could determine which cells treasured a given memory, but we didn't know how this happened. Robert Malenka of Stanford University and his colleagues discovered that increasing the amount of CREB in certain neurons makes them more easily activated. Could this increase in activity be why neurons with higher levels of CREB are selected to store memories?

To answer this question, Zhu engineered neurons in the amygdala to produce more CREB. Using tiny microelectrodes, she tested how easily these neurons are activated. The results confirmed that the engineered neurons are activated more easily compared to unmodified neurons. The high level of activity (meaning an increased chance of receiving and transmitting electrical signals carrying information between the nerve cells) implied that the cells are more adapted to launch the series of processes essential to consolidate a memory.
To test the idea, Zhu also examined the synaptic connections of the neurons with the high levels of CREB. A large body of evidence indicates that strengthening synaptic connections is essential for the formation of memories. After the mice were trained to perform a task that evoked emotional memories, she tested the strength of the synaptic connections between neurons in the amygdala that contained high levels of CREB, to see if they had stronger connections compared to cells that had not been engineered to express more CREB. To do so, she stimulated the synapses of these cells with a small electric current and recorded the responses with the help of tiny electrodes that were inserted into the cells. As expected, the neurons in the amygdala that contained higher levels of CREB had stronger synapses than other cells, a result consistent with the suggestion that they are more likely to store emotional memories.
In a more recent study, Joslin's lab showed that a memory of a fearful event can be stored in a predetermined set of neurons in the amygdala by engineering them to express a specific type of ion channel that increases the neurons' activity level. Ion channels create holes on the surface of cells, and the channels Jocelyn chose allow cells to operate more easily. Similarly, the laboratory of neurobiologist Albert Lee at the Genelia Research Campus of the Howard Hughes Medical Institute in Ashburn, Virginia, reported that artificially increasing the level of activity of neurons in a certain area of the hippocampus, while the animals ran around a running track, made neurons more likely to respond to the location The animals are on the track. This result is consistent with our findings that the level of activity plays an essential role in selecting the cells that will store a given memory.
And finally, our group, as well as Jocelyn's, took advantage of the groundbreaking technology called optogenetics, which uses light to activate or inhibit nerve cells. We used this technique to activate specific neurons with higher levels of CREB. Thomas Rogerson and Belji Jayapraksh, who were both in my lab at the time, began engineering neurons in the amygdala to make more CREB as well as ChR2, an ion channel activated by blue light. We then showed that mice could be made to recall a fearful memory by using light to activate the neurons in the amygdala that had more CREB, but not the neurons with lower levels of the protein, proof that the memory was stored in those neurons.
make connections
In 2009, I was asked to write a paper about our research on memory, and I took the opportunity to present our ideas about creating associations for memories over time. CREB's ability to identify the cells that will produce a given memory, in other words assigning memories, led me to hypothesize that this process may be an essential step in our ability to connect separate memories. My lab calls this hypothesis the "allocation of contexts" hypothesis. Because memory allocation occurs in subsets of neurons that have higher levels of CREB and are more easily activated, this process increases the probability that these neurons will store additional memory. When two memories have many neurons in common, they are actually linked together.
As a result, the activation of these neurons during the recall of one memory causes the recall of another. The key point in this idea was the expectation that two memories formed close in time, both on the same day, would be more likely to be related to each other than when they are separated by longer periods of time. When the time intervals are much longer than a day, the second memory is no longer affected by the increase in the level of activity caused by the first memory, and therefore it is consolidated in a different group of nerve cells. The time limit for making connections between memories makes sense because there is a greater chance that there is a connection between events that occur within the span of a day than between events that occur within the span of a week, for example.
Writing the article and clarifying these ideas challenged me even more to try to test them. The context allocation hypothesis is quite simple, but it was not at all clear how to test its correctness. I had to wait for the right time. This situation changed when Dennis J. Kaye and Justin Schaube, both then in my lab, joined the research. Kai thought of a clever idea. Together with Schwab, she exposed mice to two cells during the same day within a period of five hours, hoping that the memories from the two cells would be linked together. She then gave them a mild electric shock to the foot in the other cell. As expected, when she then placed the mice in the chamber where they received the shock, they froze, presumably because they remembered that's where they received the electric shock. Mice freeze in a natural response to fear because most predators are good at spotting prey when it moves.
A key prediction of our hypothesis was that separate memories formed at close intervals of time are stored in the same brain region in overlapping populations of neurons.
The important result was obtained when Kai and Schwab placed the mice in the neutral chamber. We assumed that if the memories from the two cells were linked to each other, the mice in the neutral space would remember the electric shock in the other cell and therefore freeze in anticipation, and this is indeed what happened. We also hypothesized that the probability of linking the memories would decrease if they were separated by seven days. Indeed, re-exposing the animals to the neutral chamber after a longer period of time did not remind them of the electric shock chamber and they did not freeze. In general, when the periods of time were much longer than a day, the memories were not linked together.
These behavioral findings were exciting, but they did not test the main prediction of the hypothesis - namely that separate memories formed in close time periods are stored in the same brain region in an overlapping population of neurons. This physical overlap links two memories, so that recalling one also recalls the other.
see memories
To really test the context assignment hypothesis it is necessary to actually see the memories in the brain as they are formed. There are already methods for imaging nerve cells in live mice, but they all involve fixing the mouse's head to a large microscope, which does not allow conducting the behavioral experiments needed to test the hypothesis.
It's amazing to me how many times during my career the right method has been developed just when we needed it the most. I happened upon a lecture at the University of California in Los Angeles, by Mark Schnitzer from Stanford, who described a tiny microscope that his laboratory had just invented, through which the activity of nerve cells in freely moving mice could be observed. This microscope, which weighs two to three grams, can be worn like a hat on the animal's head. This device is what we needed to track the neurons that are activated by a given memory. It allowed us to determine whether the same neurons are activated several hours later while forming another memory, a key prediction of the context assignment hypothesis.
We were so excited by the promise of this wonderful invention that we decided to build our own version of the microscope. We teamed up with the labs of Peyman Golshani and Beljit Kek, both at the University of California at Los Angeles (UCLA), and together we hired a talented postdoctoral fellow, Daniel Aharoni, who developed what we called the UCLA miniscope. Like Schnitzer's microscopes, our miniscopes were equipped with lenses that could be implanted next to the brain cells we wanted to track. The device is installed on a base that is attached to the animal's skull, and stabilizes it during the training tasks and the memory test. Just as we borrowed methods from other researchers, we were happy to share ours. We are passionate supporters of the open source movement in science and our programs and software for the UCLA miniscope are available to other groups around the world.
To observe the activity of nerve cells through the miniscope, Kai and her colleague Tristan Schuman used an imaging method that genetically engineers nerve cells in an animal so that they become alert when calcium levels in the cell rise. This method is referred to as genetically encoded calcium imaging. We decided to focus on the CA1 region of the hippocampus because of its role in learning and place memory, like the cells we used in our behavioral experiments. The mice wearing their miniscope caps were placed in both cells. We wanted to know if the time periods between the exposures to the different cells affect the selection of the nerve cells that are activated.
The results exceeded our expectations! In fact, the miniscope and behavioral experiments showed that when mice linked the memories of the two cells, many of the neurons in CA1 that were activated when the animals were in the first cell were also activated when they were in the second cell. If the time interval between the visits was about five hours, the mice formed two memories with a similar amount of nerve cells. But when the time interval increased to seven days, the pattern of overlapping activity disappeared.
We were very pleased with this finding because it confirmed a basic premise of the context allocation hypothesis: memories are linked together when they are stored in overlapping populations of neurons. If you reactivate the group of neurons of one of the two memories, it will also activate the other group and facilitate recall of the second memory.
Tagging memories
To further verify the miniscope results, Kai turned to another method developed by neurobiologist Mark Mayford, now at the University of California, San Diego. Mayford's method, known as the TetTag system (short for tetracycline tagging), was used in this experiment. When a memory is formed during a transgenic mouse's visit to a cell, the TetTag system marks active neurons with a fluorescent marker that remains intact for weeks.
In the post-mortem dissections of the animals, it is possible to compare recently activated nerve cells, marked by genes that were expressed immediately after the memory was formed, and between nerve cells marked by the long-term labeling. This move identifies not only neurons activated by one event, but also neurons activated by two events: when both types of labels glow.
Using the same experimental setup as before, Kai and her group showed that over a short period of five hours, the overlap between the neurons encoding each of the double-labeled memories was considerably greater than what would be expected by chance. In the seven-day period, the overlap between the two experiences did not rise above the level of chance.
Additional experiments by Jocelyn Betronoto's group provided further evidence for the correctness of our memory allocation hypothesis. Not only did her group conduct a different version of the nerve cell labeling experiment, but the scientists also discovered independent behavioral evidence for linking memories. The Toronto researchers hypothesized that if there is an overlap between populations of nerve cells encoding two memories, the increase in CREB level caused by the first memory will also strengthen the second memory. But instead of exposing the mice to different sites, as in our study, Jocelyn's group trained the animals to recognize two different sounds from each other. Training to recognize the first sound strengthened the memory of the second sound if both trainings occurred within six hours, but not six to 24 hours later.
Recently, Karo Inokuchi and his colleagues at Toyama University in Japan advanced the research a step further. They used optogenetics to silence the group of cells that were shared by two different emotional memories and left the other cells intact, including cells that were unique to each of the two memories. The researchers showed that by silencing the shared cells, they were able to damage the link between the two memories without affecting the ability to remember each memory separately. This elegant experiment provided direct proof that neurons shared by the two memories are essential for linking them. It also increased the number of laboratories providing independent evidence for our young memory allocation hypothesis.
Improving memory in old age
At this stage we decided to examine a link between memories in older mice. Compared to young mice, old mice have lower levels of CREB in the brain, including in neurons in the CA1 region of the hippocampus, and therefore a lower level of activity. Therefore, we believed that old mice would encounter difficulties in linking memories. Kai and her colleagues repeated many of the same experiments, this time in the old animals. The results surprised us. Experienced scientists know that hypotheses are only tools. We do not expect them to be necessarily correct. Inevitable failures help us reshape our ideas along the way. But this time, our explanation turned out to be correct.
I still remember how Kai burst into my office, a little short of breath. She told me that the older mice, although they remembered each cell individually, did have difficulty connecting the memories, even when they were exposed to them for five hours, a period of time that did not cause any difficulty for the younger mice. Compared to young adult mice, miniscope images of older animals revealed a lack of overlap between the stored memories.
Understanding how memories intertwine may help develop treatments for memory problems that characterize many psychiatric illnesses.
We were excited but also suspicious, so we immediately repeated the experiments. The second time, the results were even more convincing. The neurons in the old mice with the low CREB levels did not associate memories as easily as young mice.
The results encouraged us to expand the scope of our research. Could we artificially increase the activity level of a subset of neurons in CA1 just as the older mice explore both cells, and thereby ensure that some of the CA1 neurons that were activated in one cell will also be activated when the animals move in the other cell?
To achieve this, we took advantage of a breakthrough method known as DREADD which involves the genetic engineering of receptors on the surface of the cell, and allows the cell's activity level to be controlled. Activation of the DREADD receptors allows us to activate the same subset of CA1 neurons while the animals explore the two cells, thereby linking the memories of the two complexes.
I must confess that the idea of this experiment sounds far-fetched. There are numerous reasons why it should fail. First, memories of places involve the activation of millions of neurons spread across a large number of interconnected brain regions, and not just in the CA1 region. Older age could have affected memory binding in most, if not all, of these areas. Thus, even if we succeed in increasing the activity level of a subset of neurons in CA1, they may not be the correct cells. Moreover, we may not be able to stimulate the right level of activity.
But the experiment was successful. The key to the success of such experiments with low chances of success is to balance the investment of time and money with the possible return. However, in this case I can confidently say that luck was on our side. By reproducing the increase in the level of activity in a specific subset of neurons in the CA1 of middle-aged mice, we were able to assign two memories to many of the same cells in the CA1 and thus restore the ability to link memories in the older mice.
Studies from other laboratories conducted in both rodents and humans have also shown how it is possible to weave one memory into another. Neurobiologist Howard Eichenbaum of Boston University showed that rats are able to associate memories that have common content. Neurobiologist Alison Preston of the University of Texas at Austin and her colleagues have shown that when memories have common content, people associate them more easily. Remembering one will most likely lead to remembering the other.
The growing toolbox at our disposal to measure and control neural activity is beginning to unravel the mechanisms our brains use to organize information. Our group is now trying to extend this research in new ways. In collaboration with the computational neurobiologist Peniota Fiorzi, from the Institute of Molecular Biology and Biotechnology at the Hellas Institute of Research and Technology in Greece, we are developing computer simulations of how memories are linked together. We are also trying to decipher the mechanisms that control the time periods required to enable linking of memories in different brain structures.
So far, several large-scale experiments conducted in different laboratories provide strong support for the memory allocation hypothesis. We hope that understanding how memories intertwine will help us develop treatments for common memory problems in a variety of psychiatric conditions, from age-related cognitive decline and schizophrenia, to depression and bipolar illness. Apart from the clinical implications, the studies we have described reflect a new and exciting era in memory research, where the experiments we conduct are no longer limited by the methods at our disposal but by the power of our imagination.
