An unexpected connection was discovered between the nervous system in the body and the pancreas causing diseases accompanied by pain and disability. It turns out that they can be treated with a "pacemaker". It turns out that the nervous system is connected to vital organs and sensory organs and they also shape our health. If we learn to control them, the future of medicine will be electric.
By Gaia Vince, Mosaic Science website
When Maria Freind, a former gymnast from Volendam in the Netherlands discovered that the only way she could roll socks in the morning was to lie on her back with her feet in the air, she had to admit something was up. "I was so stiff and I couldn't get up" she says. "It was a big shock, because I'm an active person."
It was 1993. Frind was then in her late forties, working two jobs: as an athletic trainer and as a caregiver for people with disabilities, but now the physical problems began to take over her life. "I had to stop my jobs and realize that I had become disabled myself."
By the time she was diagnosed, seven years later, she was in severe pain and could no longer walk. The knees, ankles, wrists, elbows and shoulder joints were hot and inflamed. Only after years of suffering was she diagnosed with rheumatoid arthritis, a common but incurable autoimmune disease in which the body attacks its own cells, in this case the cells of the synovial joint, causing chronic inflammation and bone deformity.
The waiting rooms outside the RA clinics were full of people in wheelchairs. This sight is rare today thanks to a new wave of new types of drugs - biological drugs - highly targeted, genetically modified proteins that really help. However, these drugs do not help all patients feel better. Even in countries with the best healthcare systems, at least fifty percent of patients continue to suffer from symptoms.
Like many patients, Friend received various medications, including pain relievers, a cancer drug called methotrexate that slows down the entire immune system, and biological drugs that block the production of specific inflammatory proteins. The drugs made it easier for her, until suddenly, one day in 2011, they stopped working.
"I was on vacation with my family and my joints hurt a lot, and I couldn't walk. My daughter-in-law had to carry me.” Friend explains. She was rushed to a hospital, where she was connected to an IV and given another anti-cancer drug that targets her white blood cells. "It helped," she admits, but she worried about long-term reliance on this type of medication.
Fortunately, she doesn't have to. When she prepared herself for a life of disability and chemotherapy treatments at least once a month, a new treatment was developed that should challenge the understanding of how the interaction between the brain and the body works, in the process that controls our immune system. This is a completely new approach to the treatment of rheumatoid arthritis and other autoimmune diseases, which will make it possible to heal inflammation through the nervous system. This may even lead to research on how we might use our minds to cure disease.
And like many good ideas, this one came from an unexpected source.
The Sadness Hunter
Kevin Tracy, a neurosurgeon from New York decided to become a doctor and researcher following a wave of tragic events in his childhood. "My mother died of a brain tumor when I was five years old. It was very sudden and unexpected," he says. "I realized that the brain and nerves are responsible for health," he said. This experience led to his decision to become a brain surgeon. Then, during his training at the hospital, he came across a patient with severe burns who suddenly had severe inflammation. "She was an 11-month-old baby named Janice and she died in my arms."
These traumatic events made him think a lot about inflammation. This incident enlightened him with a new point of view that allowed him to interpret the results of an experiment in a new way.
In the late nineties, Tracy conducted experiments on the brain of a rat. "We injected an anti-inflammatory drug into the brain to study the effect of inflammatory blockage of the brain during a stroke," he recalled. "We were surprised to find that when the drug was present in the brain, it caused the spleen to release anti-inflammatory substances to other organs in the body. This despite the fact that the amount of the substance we injected was too small to pass through the bloodstream to the rest of the body."
After months of struggling, he finally came up with the idea that the brain might use the nervous system - specifically the vagus nerve - to get the spleen to turn off inflammation anywhere in the body.
(Editor's note, according to Wikipedia, the vagus nerve or vagus nerve is the tenth pair of cranial nerves. Its main function is to innervate internal organs in the body as part of the parasympathetic nervous system. About 80%-90% of the fibers in the vagus nerve are used for sensation from those internal organs in the body.)
It was an unusual idea: when a person's health is normal, and inflammation breaks out in some body tissue, it would take care of itself through the brain. Communication between the cells of the immune system that specialize in our organs and blood vessels, and the electrical connections of the nervous system was considered impossible. Tracy now discovered that these two systems were close and interwoven.
The first critical test of this exciting hypothesis was when they cut the vagus nerve in rats. When Tracy and his team injected the anti-inflammatory drug into the brain of a rat whose vagus nerve had been severed, it no longer affected the rest of the body. The second test was to stimulate the nerve without injecting any drug. "Since the vagus nerve, like all nerves, transmits information through electrical signals, this means that we should be able to replicate the experiment by placing a nerve stimulator on the vagus nerve in the brain stem and use it to cause the spleen to block inflammation.", he explains. "That's what we did and it was the experiment that led to the breakthrough."
The wandering nerve
The vagus nerve begins in the brainstem, just behind the ears. It continues down each side of the neck, chest and abdomen. VAGUS, its medical name means 'wandering' in Latin, and indeed this bundle of nerve fibers reaches the entire body, and connects the brain with the stomach and digestive system, lungs, heart, spleen, intestine, liver and kidneys, not to mention a variety of senses involved in speech, eye contact, in facial expressions and even in our ability to listen to other people's voices. It is made of thousands of fibers, 80% of which are sensory, meaning they report through the vagus nerve back to the brain what is happening in the other organs of the body.
Ups and downs in the nervous system
Although it operates far below the threshold of our consciousness, the vagus nerve is essential to maintaining our body's health. It is an essential part of the parasympathetic nervous system, which is responsible for relaxing organs after the body's response to a "fight or flight" type of danger in which adrenaline is secreted. However, not all vagus nerves are the same. Activity There are some people who have a strong vagus nerve that causes their body to relax faster after stress."
The strength of the response of the vagus nerve is known as vagal tone and can be measured using an EKG device that measures the heart rate. Every time we breathe, the heart beats faster to speed up the flow of oxygen around the body. When exhaling, the heart rate decreases. This variability is one of many features of the body controlled by the vagus nerve, which is active during exhalation but suppressed during inhalation, and thus the greater the difference between the heart rate during inhalation and exhalation, the greater the vagal tone.
Studies have shown that a high vagal tone improves the regulation of blood sugar levels, reduces the risk of diabetes, stroke and cardiovascular diseases. Low vagal tone, on the other hand, is associated with chronic inflammation. As part of the immune system, inflammation has a useful role in helping the body heal after an injury, for example, but over time it can cause damage to organs and blood vessels. One of the functions of the vagus nerve is to reset the immune system and turn off the production of proteins that cause inflammation when the role of inflammation is over. In people with low vagal tone, this control is less effective and the inflammation can spread to excessive proportions, as in the case of Maria Friend's rheumatoid arthritis, or in toxic shock syndrome, which Kevin Tracy believes caused the death of little Janice.
There is evidence that the vagus nerve plays a role in a variety of chronic inflammatory diseases, including rheumatoid arthritis. Tracey and his colleagues wanted to see if interfering with its function could serve as a possible route to treatment. The vagus nerve works as a two-way messenger, transmitting electrochemical signals between the organs and the brain. In chronic inflammatory disease, Tracy realized, the messages from the brain instructing the spleen to turn off production of a certain inflammatory protein, (TNF), were not sent. Maybe the signal transition can be improved?
Tracy spent the next decade meticulously mapping all the neural pathways involved in TNF regulation, from the brainstem to the mitochondria inside all our cells. Eventually, having gained insight into the degree to which the vagus nerve controls the dialectic response, Tracy was ready to test whether it was possible to interfere with vagus nerve activity to cure disease in humans.
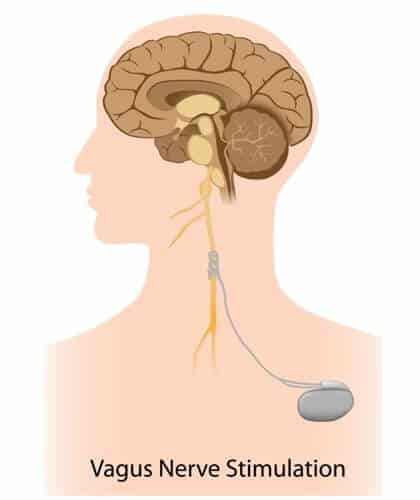
Stimulation experiment
In the summer of 2011, Maria Friend saw a newspaper ad calling for people with severe rheumatoid arthritis to volunteer for a clinical trial. The participation involved the insertion of an electrical implant connected directly to the vagus nerve. "I called them immediately," she says. “I don't want to take cancer drugs all my life; It's bad for your organs and dangerous in the long run. "
Tracy designed the trial with his partner, Paul-Peter Tuck, professor of rheumatology at the University of Amsterdam. Tuck has long been looking for an alternative to powerful drugs that suppress the immune system to treat rheumatoid arthritis. "The body's immune response becomes a problem when it attacks its own cells and not foreign cells, or when the inflammation becomes chronic," he reasons. "The question becomes: how can we improve the body's shutdown mechanism? How can we achieve results? "
When Tracy called him out and hypothesized that stimulating the vagus nerve might be the cause of shutting down TNF production, Tuck quickly saw the potential and was curious to see if it would work. Stimulation of the vagus nerve has already been approved in humans for epilepsy, so it will be relatively simple to get approval for a trial in arthritis. One of the obstacles they faced was the question of whether people who are used to taking medication for their condition, would be willing to undergo surgery to implant a device inside their body. "We didn't know if patients would agree to receive the neuro-electrical device like they receive a pacemaker" says Tuck.
He needn't have worried. Over a thousand people expressed interest in the procedure, far more than were necessary for the experiment. In November 2011, Friend was the first of twenty Dutch patients on whom the device was tried.
"They put the pacemaker in the left side of my chest, with wires coming out of it and joining the vagus nerve in my throat," she says. "We waited two weeks for the area to heal, and then the doctors activated the pacemaker and adjusted the settings for me."
She received a magnet and was required to wear it around her neck six times a day. The magnet activated the implant, causing the vagus nerve to be stimulated for thirty seconds at a time. The hope was that this would reduce her inflammatory response. When Friend and another trial participant were sent home, it was a waiting game for Tracy, Tuck and the staff who wanted to see if the theory, laboratory studies and animal experiments would bear fruit in real patients. "We hoped that some patients would benefit from the relief of their symptoms, maybe their joints would hurt a little less," says Tuck.
At first Friend was too eager for the wonder drug. She immediately stopped taking her pills, but her symptoms returned so badly that she was bedridden in excruciating pain. She went back on the pills and they were tapered over a week.
Then something extraordinary happened. Friend experienced a more impressive recovery than she or the scientists dared hope for.
"Within a few weeks, I was in great shape," she says. "I could walk again, I went back to ice skating and my gymnastics. I feel much better. She is still required to take low-dose methotrexate for the rest of her life, but Fried, a 68-year-old retiree, now plays and teaches groups of seniors to play volleyball, does gymnastics and plays with her eight grandchildren.
Other patients in the trial had similar experiences of sharp improvement. The results aren't ready for publication yet, but Tuck says more than half of the patients showed significant improvement and about a third saw remission of their rheumatoid arthritis. Not only did sixteen of the 20 patients in the trial feel better, but the level of inflammation in their blood also decreased. Some now take no medication at all. Even those who did not experience significant clinical improvements from the implant insisted that it helped them; No one wanted it removed.
"We showed good results when we activated the stimulation for three minutes a day," says Tuck. "When we stopped stimulation, we saw that the disease returned and the levels of TNF in the blood increased. When we woke up, the body returned to normal."
Tuck suspects that patients will continue to need vagus nerve stimulation for life. But unlike the drugs, which work by preventing the production of immune system cells and proteins such as TNF, stimulating the vagus nerve seems to restore the body's natural balance. The stimulation reduces the overproduction of the TNF that causes chronic inflammation but does not affect the function of the healthy immune system, so the body can respond normally to the infection.
"I'm really glad that I participated in the experiment," says Friend. "Over three years have passed and my symptoms have not returned. At first I felt pain in my head and throat when I used the implant, but within a few days it stopped. Now I don't feel anything but a constriction in my throat and a tremor in my voice while it works."
"I occasionally have stiffness or a little pain in my knee, but these symptoms disappear within a few hours. I don't have any side effects from the implant, like I had from the medication, and the effect doesn't go away like when it happens after taking medication."
Having an electrical device surgically implanted in your neck for the rest of your life is a serious procedure. But the technique proved so successful at this and so attractive to patients that other researchers decided to try using vagus nerve stimulation for a wide range of other serious chronic diseases, including inflammatory bowel disease, asthma, diabetes, chronic fatigue syndrome and obesity.
And Maria Friend, despite severe rheumatoid arthritis can now walk around her picturesque village without pain and has a new life: "It's not a miracle - the researchers told me how it works using electrical impulses - but it feels magical. I don't want to ever have my device removed. I got my life back! "
Translation: Avi Blizovsky
To the article on the Mosic Science website
More of the topic in Hayadan:
- Transport of molecules in the nervous system
- How does a gene "sculpt" neurons?
- Researchers have succeeded in creating an interface between the nervous system near a prosthesis

8 תגובות
The strength of the response of the vagus nerve is known as vagal tone and can be measured using an EKG device that measures the heart rate. Every time we breathe, the heart beats faster to speed up the flow of oxygen around the body. When exhaling, the heart rate decreases. This variability is one of many features of the body controlled by the vagus nerve, which is active during exhalation but suppressed during inhalation, and thus the greater the difference between the heart rate during inhalation and exhalation, the greater the vagal tone.
I wanted to ask if it is possible to explain in other words or in more detail the meaning of the last sentence.
Thanks
Rohik
The method is known and is called vagus nerve stimulation - VNS. There are also articles on the relationship between vns and Chinese medicine (for example: Neuroanatomic and Clinical Correspondences: Acupuncture and Vagus Nerve Stimulation).
The fact that inflammation can be regulated through nerve stimulation is fascinating and has great interest in understanding the way Chinese medicine works, not only in inflammation but also in pathologies such as depression. Benny Aaron from the comment above, you are absolutely right in the link you made, but there are studies and there is no doubt that there is much more to explore.
Ahh
Sons of Aaron
Who does not give opportunities for research? In the study above, they showed us an understanding of the mechanism followed by a scientific examination of the treatment - the use of statistics including reviews.
Do even just a statistical study of testing the effectiveness of acupuncture, but properly, with the right reviews and the placebo and guaranteeing you that no one will have a problem receiving it. If you manage to understand the mechanism at all you have played it.
The only requirement is that the research be scientific, that is, meet certain standards. "Natural" medicine, as you call it, currently does not meet these standards. Do you know what natural medicine is called that does meet these standards? medicine.
Good article. Thanks. And we hope that the patients could benefit from this development soon.
And for Beni Aharon, I am in favor of natural (alternative) medicine and always have been, but it doesn't always help either. And here we are talking about implants inside the body, which probably help, and for that traditional medicine is necessary.
Bnei Aharon, what is natural medicine? Is supernatural medicine included in the framework of natural medicine and on the basis of what information do you determine that research is being carried out on alternative medicine {a more appropriate name, although in my opinion it is advisable to replace the word medicine with a concept, way, information or something like that, since it is bigger than medicine, it is about A completely different way of life, a way that denies science as a means of understanding the world and does not perceive the information in the world as incremental.
We know it from natural medicine such as acupuncture, etc.
It's a shame that we are not given opportunities for research unfortunately
exciting