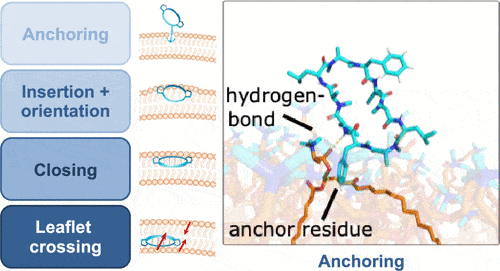
First, the molecule anchors itself to the surface of the membrane, before it penetrates the membrane in a direction perpendicular to it. In the next step, it changes its three-dimensional structure while passing through the membrane, rotating once along the longitudinal axis before reaching the opposite side of the membrane, where it exits the membrane again
[Translation by Dr. Moshe Nachmani]
There is a constant need for new drugs. For example, many of the antibacterial drugs (antibiotics) that we have been using for a long time develop resistance and become less and less effective. Chemists and pharmaceutical scientists are frantically searching for new active ingredients, especially those that can penetrate cell membranes, since these are the only types that patients can take orally (by mouth) in tablet or syrup form. Only these active substances can pass through the walls of the small intestine and enter the bloodstream in order to reach the required area of the body. For active substances that are unable to penetrate through the cell membrane, doctors have no choice but to inject them directly into the bloodstream
Large compounds with great capacity
This is the reason why scientists try to understand in advance what compounds will be able to penetrate through the cell membrane and exactly how they do so. For such an important and promising family of substances - cyclic peptides - chemists from the University of Zurich (ETH Zurich) were able to gain new insights into the relevant mechanism. "The more we know about this mechanism and about the properties that such compounds must exhibit, the earlier and more effectively researchers will be able to take this data into account when developing new drugs," says Serina Riniker, a professor in the Department of Chemistry and Applied Biosciences. She led the research that has just been published in the scientific journal Journal of Medicinal Chemistry.
Cyclic peptides are ring-shaped molecules, larger than the small molecules that make up most drugs today. In several fields of application, on the other hand, chemists and pharmaceutical scientists struggle hard with the limitations of these small molecules and instead turn to excessively large molecules such as cyclic peptides. This family of substances includes many natural substances with pharmaceutical activity, such as cyclosporine, which is an immunosuppressive drug used to treat diseases and conditions in which immunosuppression is required, such as autoimmune diseases or after organ transplants, to prevent transplant rejection, as well as antibacterial drugs many
The use of computer modeling
With the help of computer modeling and the great power of supercomputers, the researcher and her colleagues were able to explain how cyclic peptides similar to cyclosporine manage to penetrate through the cell membrane. "Only modeling allows us to get such great details, insights with high resolution, because there is no experiment that allows us to observe a single molecule passing through the membrane," explains the lead researcher. To understand the mechanism we must first know how cyclic peptides are built: they consist of a central ring structure to which side groups are attached. These molecules are flexible and are able to dynamically change their structure in order to adapt to their environment.
Dance through the cell membrane
The researchers' imaging revealed how a cyclic peptide penetrates through the membrane: first, the molecule anchors itself to the surface of the membrane, before penetrating the membrane in a direction perpendicular to it. In the next step, it changes its three-dimensional structure while passing through the membrane, rotating once along the longitudinal axis before reaching the reverse side of the membrane, where it exits the membrane again. These changes in the spatial structure of the molecule result from different environments that the molecule experiences while passing through the membrane - the body is mainly composed of water. Both inside and outside cells, most biochemical molecules are in aqueous solution. Cell membranes, on the other hand, are composed of fatty acids, so hydrophobic conditions dominate the content. "In order to allow it to cross the membrane, the ring peptide changes its spatial shape rapidly in order to become as hydrophobic (water repellent) as possible," said the lead researcher.
Changing the molecular side chains
In the current study, the researchers examined eight different types of cyclic peptides. These are model peptides without any medical effect that the same pharmaceutical scientists from the giant corporation Novartis developed for fundamental research, a fact that explains why the main researcher collaborated with this corporation in this study. The new findings can now be used in the field of the discovery of ring peptides as candidates for new drugs. At the same time, the researcher points to a certain compromise: there are side chains that provide optimal conditions for anchoring to the surface of the membrane, but this very fact makes it difficult for the peptides to pass through the membrane.
This cutting-edge knowledge is helping researchers move toward understanding which side chains will be best suited for use. These findings could speed up drug discovery by revealing the most suitable conditions for the development of active substances that could eventually reach the body in tablet form.

One response
It is written that the drugs develop resistance. Is it not the bacterium or the virus that develops the resistance? In addition to this, the description implies that the molecule has a "will", isn't it more correct to write that the components of the chemical environment allow the molecule to attach or move, etc.?